|
With the combined results of Thomson and Millikan, a value for the electron mass was obtained - a value far below that of atoms. The determination of the charge of the electron awaited the work of Millikan who measured the electron charge in 1909 with his oil drop experiment. Using this apparatus, Thomson determined the charge-to-mass ratio of the electron, e/m. This same principle is presently used in velocity selectors for mass spectrometers. Thomson showed that with the application of both electric and magnetic fields, he could balance the deflections and obtain a straight beam. A narrow luminous beam could be produced by using an aperture near the cathode, and this beam could be deflected by either an electric field or a magnetic field. In the 1890's, cathode ray tubes had been developed in which a luminous beam could be produced in a partially evacuated glass tube, directed from the negative electrode (cathode) to the positive (anode). Sir Joseph John Thomson (1856-1940) played a pivotal role in developing our understanding of the electron. Thomson was able to apply electric and magnetic fields to manipulate the rays, which eventually convinced the physics world that they were composed of tiny particles, electrons, opposed to waves in the now-rejected ether.įind out more about Thomson and the story of the first subatomic particle here, or visit the Museum to see Thomson’s cathode-ray tube in the Collider exhibition. If you’re interested in the details of how Thomson and Everett conducted their experiments visit the Cavendish Lab’s outreach page here.J. Only when almost all the air has been removed to create a high vacuum – a state that would shatter ordinary glass vessels – can the rays travel the full length of the tube without bumping into air molecules. The quality of Everett’s glassblowing was absolutely crucial for the experiments to work.Ĭathode-rays are produced when an electric current is passed through a vacuum tube. Everett made all of Thomson’s apparatus, and was responsible for operating it – in fact, he generally forbade Thomson from touching anything delicate on the grounds that he was “exceptionally helpless with his hands”. Cambridge’s Cavendish Laboratory, where Thomson spent his scientific career, also has an original tube in its collection.Įach tube was custom-made by Thomson’s talented assistant, Ebenezer Everett, a self-taught glassblower. Using more than one cathode-ray tube in 1897 for his experiments, Thomson managed to identify a particle 1,000 times smaller than the then known smallest piece of matter: a hydrogen atom. I had read lots about Thomson’s famous experiments on the electron – the first subatomic particle to be discovered – but to actually see and touch his apparatus myself, to notice the blackened glass and the tube’s minute features that are omitted in books, brought the object to life.  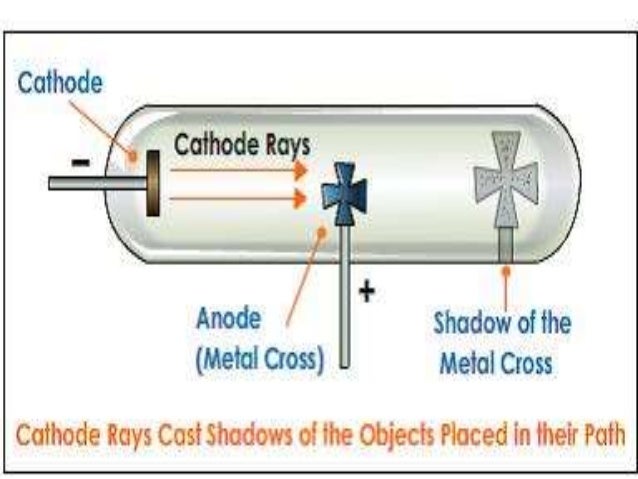
Holding the delicate glass cathode-ray tube in my hands, once used by the great physicist JJ Thomson, was an incredible treat, and an experience I will never forget. Rupert Cole celebrates JJ Thomson’s birthday with a look at one of the star objects in our Collider exhibition. 
Rupert Cole celebrates JJ Thomson's birthday with a look at one of the star objects in our Collider exhibition.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
